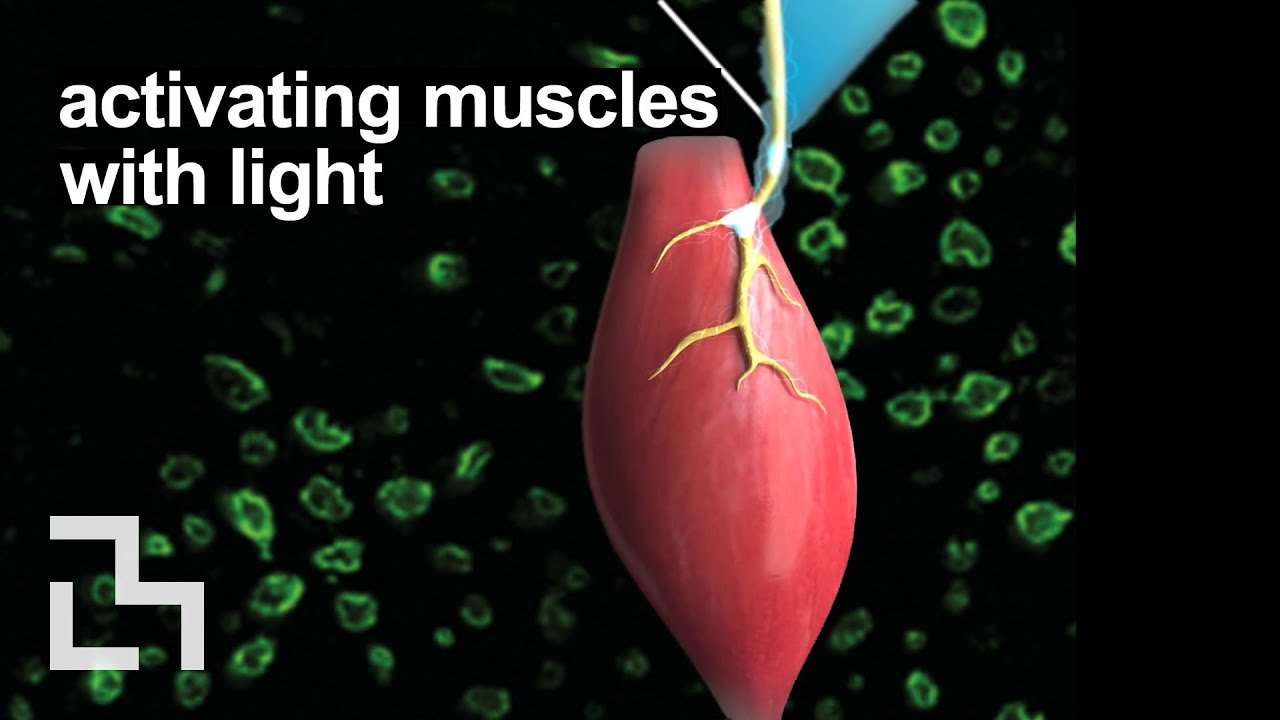
For people with paralysis or amputation, neuroprosthetic systems that artificially stimulate muscle contraction with electrical current can help them regain limb function. However, despite many years of research, this type of prosthesis is not widely used because it leads to rapid muscle fatigue and poor control.
MIT researchers have developed a new approach that they hope could someday offer better muscle control with less fatigue. Instead of using electricity to stimulate muscles, they used light. In a study in mice, the researchers showed that this optogenetic technique offers more precise muscle control, along with a dramatic decrease in fatigue.
“It turns out that by using light, through optogenetics, one can control muscle more naturally. In terms of clinical application, this type of interface could have very broad utility,” says Hugh Herr, a professor of media arts and sciences, co-director of the K. Lisa Yang Center for Bionics at MIT, and an associate member of MIT’s McGovern Institute for Brain Research.
Optogenetics is a method based on genetically engineering cells to express light-sensitive proteins, which allows researchers to control activity of those cells by exposing them to light. This approach is currently not feasible in humans, but Herr, MIT graduate student Guillermo Herrera-Arcos, and their colleagues at the K. Lisa Yang Center for Bionics are now working on ways to deliver light-sensitive proteins safely and effectively into human tissue.
Herr is the senior author of the study, which appears today in Science Robotics. Herrera-Arcos is the lead author of the paper.
Optogenetic control
For decades, researchers have been exploring the use of functional electrical stimulation (FES) to control muscles in the body. This method involves implanting electrodes that stimulate nerve fibers, causing a muscle to contract. However, this stimulation tends to activate the entire muscle at once, which is not the way that the human body naturally controls muscle contraction.
“Humans have this incredible control fidelity that is achieved by a natural recruitment of the muscle, where small motor units, then moderate-sized, then large motor units are recruited, in that order, as signal strength is increased,” Herr says. “With FES, when you artificially blast the muscle with electricity, the largest units are recruited first. So, as you increase signal, you get no force at the beginning, and then suddenly you get too much force.”
This large force not only makes it harder to achieve fine muscle control, it also wears out the muscle quickly, within five or 10 minutes.
The MIT team wanted to see if they could replace that entire interface with something different. Instead of electrodes, they decided to try controlling muscle contraction using optical molecular machines via optogenetics.
Using mice as an animal model, the researchers compared the amount of muscle force they could generate using the traditional FES approach with forces generated by their optogenetic method. For the optogenetic studies, they used mice that had already been genetically engineered to express a light-sensitive protein called channelrhodopsin-2. They implanted a small light source near the tibial nerve, which controls muscles of the lower leg.
The researchers measured muscle force as they gradually increased the amount of light stimulation, and found that, unlike FES stimulation, optogenetic control produced a steady, gradual increase in contraction of the muscle.
“As we change the optical stimulation that we deliver to the nerve, we can proportionally, in an almost linear way, control the force of the muscle. This is similar to how the signals from our brain control our muscles. Because of this, it becomes easier to control the muscle compared with electrical stimulation,” Herrera-Arcos says.
Fatigue resistance
Using data from those experiments, the researchers created a mathematical model of optogenetic muscle control. This model relates the amount of light going into the system to the output of the muscle (how much force is generated).
This mathematical model allowed the researchers to design a closed-loop controller. In this type of system, the controller delivers a stimulatory signal, and after the muscle contracts, a sensor can detect how much force the muscle is exerting. This information is sent back to the controller, which calculates if, and how much, the light stimulation needs to be adjusted to reach the desired force.
Using this type of control, the researchers found that muscles could be stimulated for more than an hour before fatiguing, while muscles became fatigued after only 15 minutes using FES stimulation.
One hurdle the researchers are now working to overcome is how to safely deliver light-sensitive proteins into human tissue. Several years ago, Herr’s lab reported that in rats, these proteins can trigger an immune response that inactivates the proteins and could also lead to muscle atrophy and cell death.
“A key objective of the K. Lisa Yang Center for Bionics is to solve that problem,” Herr says. “A multipronged effort is underway to design new light-sensitive proteins, and strategies to deliver them, without triggering an immune response.”
As additional steps toward reaching human patients, Herr’s lab is also working on new sensors that can be used to measure muscle force and length, as well as new ways to implant the light source. If successful, the researchers hope their strategy could benefit people who have experienced strokes, limb amputation, and spinal cord injuries, as well as others who have impaired ability to control their limbs.
“This could lead to a minimally invasive strategy that would change the game in terms of clinical care for persons suffering from limb pathology,” Herr says.
The research was funded by the K. Lisa Yang Center for Bionics at MIT.